We Find SRB Deep Inside the Bolt
The fourth part of a multi-part series
In conducting this work I have been greatly assisted by Luis Fernandes Silva and Rui Rosado who have provided multiple samples and photos for analysis. Luis has spent a great deal of time measuring spot-sulphate levels, and the pH profiles of bolts he has extracted during route refurbishment. We now know a lot more about why the Cabo da Roca crags eat stainless steel.
1. Introduction:
In Part 3 of this series we started out on the long journey to find an explanation for the brittle fracture that plagues the bolted infrastructure installed on the sea-cliffs of Cabo da Roca. In that earlier post, I summarised the phenomenon of hydrogen embrittlement (HE) as it presents at the macroscopic level. I deliberately left the atomistic viewpoint as a subject for later discussion.
This current post is something of a side-step, in that we will review the rather remarkable finding that sulphate reducing bacteria (SRB) are anything but superficial in their action, and these tiny organisms can penetrate the bulk material of the bolt to a significant degree. The importance of this finding is two fold. Firstly there is the issue of weakening the material by the direct corrosion of bulk metal, and secondly, with bacteria distributed within the heart of the bolt, it becomes ever more likely that significant hydrogen levels might accrue.
To be clear, this is the phenomenon we are addressing.
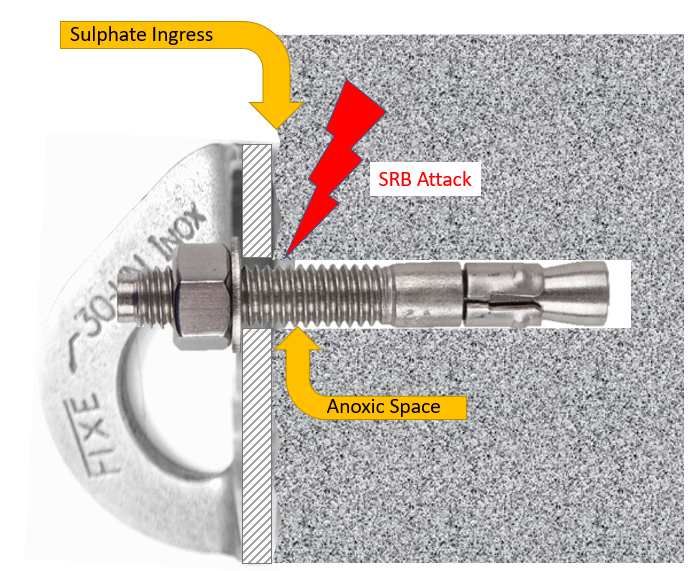
Up to this point, my working theory had been that SRB would occupy just the anoxic zone between bolt and rock, and from that location would facilitate hydrogen entry into the bulk metal by sponsoring a cathodic charging process at the surface of the bolt.
However, I will present evidence that SRB themselves are entering the metal by some mechanism, perhaps mixture of mechanisms, and that the wide distribution of sulphide throughout the bolt is the direct result of their presence within the metal itself. It would seem that these micro-organisms are colonising the solid metal of the bolt.
As I have argued elsewhere, it is perfectly feasible for atomic hydrogen to slowly diffuse into a piece of 304, but, for the sulphide ion, surely never. Well, never is too strong a word. When I searched the literature, I did locate one reference to sulphide diffusion through a copper alloy , but no, let’s scrap that esoteric idea while there are simpler, more plausible, mechanisms available to explain what we are seeing.
2. Distribution of Metal Sulphide:
One of the more unnerving things about the brittle fracture phenomenon is the fact that dangerously brittle bolts often exhibit minimal signs of corrosion, and therefore can appear fit for use. Indeed, it is only when we look closely at the microscopic level do we begin to see the tell-tale signs of SRB activity, with penetrative corrosion features, and deposits of various iron sulphides.
If this lack of superficial corrosion is surprising, then it is nothing compared with the quite unexpected finding that all such compromised bolts exhibit sulphide deposits deep within the bulk of the metal.
I should emphasize that this accumulation of internal sulphide is something that happens during the service period of the bolt. In comparison, the sulphide content of a newly installed 304 bolt is very low, and is less than 0.03% S by specification.
The most striking evidence we have for the distribution of metal sulphide is the reaction that occurs when the iodine-azide reagent is applied to the fracture surface. If a solid surface exposes sulphide at any point, then bubbles of nitrogen gas will be seen nucleating around that point.
We are going to be relying on this test quite a bit, so, just how specific is it for sulphide? Well, it depends upon an unusual and quirky amplification reaction that offers a high degree of specificity. The rate of the reaction is not so much a measure of the amount of sulphide present, as it is the surface area of solid sulphide presented to the reagent. Thus a single solid crystal of metal sulphide will react quite slowly, while the same crystal ground to a fine powder will produce a rapid reaction. Very little sulphide is consumed in this process, so much so that it appears that its role is purely catalytic, but this most definitely is not the case. See for a discussion of the mechanism.
As a reference point for the resolutely sceptical, here is a picture of the reagent applied to the fractured end of a 304 bolt extracted from a benign crag, free of sulphate problems.

There is no sign of a reaction. Compare that with a bolt from Espinhaco featured in the clip below.
As I mentioned above, such a vigorous reaction does not necessarily mean a large mass percentage of sulphide is present, and it could be explained equally well by the presence of a small, but very finely distributed, amount.
Remember these six bolts from the last post?






Let’s take a closer look at that fracture.
Bolt #1: If we examine the above movie clip of the reaction that occurred at the fracture surface of bolt #1, it seems that the bubbles of nitrogen gas are being nucleated in a fairly uniform manner right across its diameter. Thus, given the nature of the iodine-azide reaction, we are forced to conclude solid metal sulphide must be distributed across the fracture surface.
Bolt #2: shows the same sort of uniform distribution across the fracture surface.

Bolt #3: tells the same story. However, in this case the surface was treated with 3.6% hydrochloric acid for 3 minutes to remove readily soluble sulphides, prior to testing with the iodine-azide reagent. The response to the reagent appears undiminished by the prior treatment.
Bolt #4: shows a lower density of nucleation points, but nevertheless illustrates the presence of sulphide in the very centre of the bolt.

Bolt #5: we find similar evidence for widely distributed sulphide.

Bolt #6: we once again observe a strong reaction to iodine-azide right across the extent of the fracture.

3. A Quick Discursion Into Iron Sulphides:
SRB have the ability to derive energy by ‘burning’ metallic iron in the presence of sulphate. That is, all they need to thrive is metallic iron and dissolved sulphate. The major product of this reaction is iron sulphide, and thus it is useful to be able to recognize the various sulphides of iron that we might come across when we examine bolts that have been subject to attack by SRB.
The biogenesis of iron sulphides is a subject of considerable breadth. provides a good starting point for those that might wish to delve into this subject. I will keep things simple by discussing just three iron sulphides, mackinawite, greigite and pyrite. I have already touched on two of these in an earlier post, and here, I’ll expand further on the differences between them.

Mackinawite
FeS = Fe^{2+}S^{2-}
iron may be partially replaced by nickel
tabular, grey, black crystals
non-magnetic
dissolves under acid conditions
auto-transforms to greigite unless anaerobic conditions prevail

Greigite
Fe_3S_4 = Fe_1^{2+}Fe_2^{3+}S_4^{2-}
iridescent dark octahedral crystals
strongly magnetic
persists under acid conditions
stable in air

Pyrite
FeS_2 = Fe^{2+}(S:S)^{2-}
metallic bronze cubic crystals
non-magnetic
persists under acid conditions
stable in air
While I make no strong claim to the accuracy of the names I’ll be assigning to the corrosion deposits discussed further down page, there are only so many possibilities for a metal sulphide. Of the common minerals above, only one is magnetic and only one is acid-soluble. Beyond these basic, testable physical properties we can be guided by literature with respect to their likely formation and relative stability.
For instance, there is abundant research to indicate that the first product of the reaction between ferrous ions and sulphide ions is mackinawite. Under neutral conditions this material will coat the iron surface with a protective film .
However, mackinawite is inherently unstable and will persist only for as long as the oxygen level is low, and a neutral pH is maintained . Traces of oxygenated species are known to catalyse the spontaneous, solid state rearrangement of the atomic lattice to that of greigite with the expulsion of two electrons per mole of the original monosulphide .
Solution phase formation of pyrite from mackinawite is also known with the expulsion of one ferrous ion per mole of mackinawite, to achieve the more stable pyrite atomic lattice . Some authors have also postulated the solid state formation of pyrite from greigite .
The Pourbaix diagram below is from . It goes some way to explaining the relationship between metal removal and the formation of the two sulphides of interest. It broadly supports the assertions I made above, but we need to remember that, as always, the real world may well confound some of the predictions we can make from this diagram.

So what do we find?:
Firstly, an almost universal finding is that of a corrosion product which resembles mackinawite, and which coats those metal surfaces protected from the atmosphere but, typically, are located toward the outside of any anchor assembly, e.g. thread flanks, between nuts and washers, and between washers and fixed hangers. This grey/black material commonly shows a distinctive, tabular textured surface, is often weakly magnetic, iodine-azide positive, soluble in 3% hydrochloric acid and spot tests for both ferrous and nickel ions. The weak magnetism I attribute to partial, spontaneous conversion to greigite once the surfaces are exposed to the air.
Secondly, and equally common is the corrosion product I attribute to greigite. It occurs as dark, iridescent deposits that exhibit triangular crystal facets and can be found, sometimes on thread flanks, but most commonly within pockets within the bulk of the metal. This material is strongly magnetic, iodine-azide positive, only poorly soluble in 3% hydrochloric acid, and tests positive for ferric ion and negative for nickel.
Thirdly, but somewhat rarely, within pockets within the bulk metal, there can be observed distinctly cubic forms which present a metallic lustre, but are much paler and quite distinct from the dark iridescent greigite. This material is resistant to 3% hydrochloric acid, but I have thus far failed to isolate a pure sample in order to test for magnetism or iodine-azide response. I’m guessing it is pyrite. I could well be wrong.
One final piece of general evidence we need to consider is the striking partitioning of pH between surfaces of an anchor under active attack by SRB. I have commented on this phenomenon in a previous post. The pH may vary from as high as pH 12 toward the outside compared with pH 2 within the structure of the anchor.


I have a very speculative explanation for the strong pH differentiation which we will come to when I consider the possible mechanisms of the SRB attack. For now the important thing to note is that we seem to find putative mackinawite in regions we know to be of elevated pH, while putative greigite shows in regions of low pH. Such a finding makes sense in terms of the Pourbaix diagram I have presented above.
4. Evidence For Penetrative Corrosion:
Armed with this knowledge of iron sulphides, we can begin to take a closer look at the fracture surfaces of our six samples.
Warning: What follows is going to get a little repetitive, but I want to establish beyond reasonable doubt the fact that brittle fracture is always accompanied by evidence that the bacteria themselves have deeply penetrated the solid metal of the bolt.
Bolt #1:
If we place the fractured end under a microscope, it doesn’t take much magnification to reveal that far from being free from corrosion, there is ample evidence of the activity of SRB within the fracture zone.
Not only are the thread flanks completely coated with what I take to be mackinawite, but we also see something that looks like greigite deposited at points well within the bulk metal the bolt.

Zooming-in a little closer it is evident that the bacteria have corroded tunnels from the thread surfaces into the metal. Points of intersection of the fracture surface with such tunnels is relatively common in the outer millimetre of the bolt.

Looking at the innermost greigite deposit we see two types of spaces being occupied by this sulphide. The first is the sharp-edged tunnel that can be seen in cross-section. The inner walls look relatively smooth as if subject to anodic dissolution of the base metal. The second is characterised by a ‘plucked’ surface as if inter-granular cohesion has failed, and voids have opened as internal stresses are relieved. It is quite likely much of this disruption was caused by the blow that fractured the bolt, but the darkened surface, taken together with the presence of greigite on that surface, points to the likely pre-existence of at least a number of fractures.



Based on the iodine-azide test we know that sulphide must be more widely distributed than just the obvious greigite deposits seen here, and thus closer inspection is warranted.
If we look carefully, it can be seen that pretty much anywhere on its surface, the apparently inter-granular fracture (we’ll challenge that notion in a future post) is interrupted by features that could well be sulphide. It is relatively easy to identify cubic, pyrite-like structures with a size that exceeds the typical grain size of the metal, and, perhaps more significantly, we can observe an abundance of zones of an amorphous grey material arranged somewhat circumferentially within the predominantly inter-granular fractured surface.

For now, I’ll do no more than note this circumferential pattern, and the apparent in-filling. In a later post, I will consider the possibility of progressive degradation of the bolt by hydrogen cracking.
Bolt #2:
This specimen presents a little differently to the one just discussed. Once again we see the thread flanks coated with mackinawite. However, the presence of greigite within corrosion tunnels is not a strong feature of this specimen.
What is striking, however, is the presence of myriad small crack-like structures, many of which appear to be infilled with a dark deposit. Those nearer the outside of the bolt (marked B) tend to be arranged somewhat circumferentially compared with those toward the centre (marked A).

This fracture exhibits the stepped structure I have previously commented on as being characteristic of the failure modes we are observing at Cabo da Roca. Such “steps” allow us to take a look at the longitudinal section.
Given that we know that the predominant stress vector in a tightened expansion bolt is going to be aligned with the longitudinal axis, it is somewhat surprising to observe a similar pattern of “filled cracks” in the longitudinal section, albeit now they tend to be somewhat longitudinally aligned.

The fact that the observed cracking is not orthogonal to the primary stress vector must carry some significance, and I’ll return to this observation in a later post.
However, greigite is present, and on closer examination it is easy enough to locate both on the outer surface, and within the small number of corrosion tunnels as illustrated below.



An intriguing question is whether those crack-like inclusions represent quantities of sulphide further to the visible greigite? Thus far, the answer to that question is proving elusive. Consider the photographs below. The initial surface showed a large number of apparently infilled cracks which present in fairly random orientation. The addition of 3.6% hydrochloric acid to this surface for one minute produced a strong odour of hydrogen sulphide, and the resultant liquid, spot-tested for just ferric ion. There was no trace of nickel or ferrous ions, so it clear the acid was hydrolysing a ferric sulphide.
Yet when the surface was washed and re-examined, it looks largely unchanged. Maybe some material has been removed from the cracks – maybe not. In any event, if sulphide has been removed, then there is no shortage of it left as is evidenced by the subsequent iodine-azide reaction.


Bolt #3:
This specimen exhibits all the features of the previous two bolts. We see mackinawite coating the thread flanks and greigite buried deep within the bulk of the metal. There are also the irregular crack features we saw with bolt #2. Some of these might well be genuine cracks resulting from the fracture, others appear to be filled.

In the photos below we see a typical presentation of greigite on the thread flanks – dark brown/red crystals tending toward iridescence, strongly attracted to a magnet and positive in the iodine-azide test.



However, as I have already outlined earlier, sulphide mineralogy is somewhat convoluted, and at it may well be that we have multiple sulphides present. For example, consider the photos below showing before and after treatment with dilute hydrochloric acid.


The white crystals are presumably soluble electrolytes such as sodium sulphate/chloride and not surprisingly are removed by the acid treatment. However, what is of interest is that we see partial attack on the putative greigite deposit. On closer examination it can be seen that some of the crystals are little more than tarnished whilst other appear to have been substantially degraded.
I wouldn’t expect greigite to be attacked, and I also wouldn’t expect to find a mono-sulphide like mackinawite within what we know to be a quite strongly acid environment. However, as I outlined earlier, there is nothing straightforward about iron sulphide mineralisation, and we could be looking at a mixed sulphide, not to mention the incorporation of other iron sulphides such as pyrrhotite.
Here is another example of something similar happening. This time it is on the longitudinal surface of a step in the fracture. Not all components of the deposit are equally vulnerable to attack by dilute acid.


Bolt #4:
This specimen differs from the others in that the level of magnetism, and thus, presumably, the strain-induced martensite content is quite low. It is of interest, therefore, to note that we see very little evidence of corrosive tunnelling and greigite deposition within the bulk metal of the bolt. There are some deposits to be seen in the longitudinal section exposed by the step fracture, but, apart from this, you need to look hard to find evidence of invasive corrosion.

Yet, there is no shortage of mackinawite coating the thread flanks. And most tellingly, there is a positive reaction to sulphide at the very centre of the fracture surface.
Bolt #5
At first glance, this bolt presents what appears to be the same brittle fracture we have seen with the other bolts discussed above. We see mackinawite coating the thread flanks, and deposits of greigite within the bulk metal. Furthermore we also observe the small irregular cracks, perhaps infilled, typical of this fracture mode.

However, upon closer examination, it is apparent that substantial hydrolysis of the putative greigite must have occurred, perhaps due to moist storage conditions., and there is a yellow brown rust-like coating in the vicinity of the dark, brown-black, semi-crystalline granules of presumptive greigite.
Given that mackinawite is more easily hydrolised than greigite, then it seems unlikely that we would see degradation of greigite but not mackinawite following storage of the fractured specimen. This fact, adds weight to the my previous observation for bolt #3, that we might have a sulphide other than pure greigite present. A mixed iron sulphide is a real possibility.





Bolt #6
This bolt is probably the best example that we have of what happens when SRB invade solid stainless steel. It exhibits three distinct zones within the fracture surface…
a) The first of these is that of a pre-existing stress-crack. It is coated with the dark, greenish-grey deposits typically associated with SRB attack. This type of deposit seems to form once the corroded surface gains partial access to the air. Some authors refer to these deposits as “green rust” . Also see “sulphate green rust” in Wikipedia.
b) The second area is free from such green surface deposits and has the appearance of a spongy, poorly-coherent, section of metal that was pulled apart, rather than fractured, when the hammer blow was applied.
c) The third section is that typical of the brittle fractures we have been considering up to this point.

We see greigite embedded in all three zones, and additionally, we also see an abundance of the small, irregular crack features we have come to recognize as typical of these fractures.
In the photo below, not only do iridescent greigite crystals coat the thread flanks, but they also are present in a number of corrosion tunnels/cracks intercepted by the brittle portion of the fracture.

Looking at the point on the surface where the pre-existing stress-crack surface gives way to brittle fracture, see photo below, we observe greigite situated within corrosion tunnels. Note the smooth margins of the tunnels cut trans-granularly through the matrix of the metal, such as we would expect for an anodic dissolution process driven by the bacteria. Look back up-page to see a very similar structure for Bolt #1.

The situation above is in direct contrast to that of the photo below. Here we examine the transition from the brittle to the spongy section of the fracture. It is not immediately obvious what is happening here. It doesn’t seem likely that the metal could be rendered to such a sponge-like consistency by anodic dissolution of tunnels through the bulk material. If such were the case, we would expect somewhat smooth trans-granular surfaces marking the tunnel walls, such as we saw in the figure above. Here the surfaces of the voids look to be exposing inter-granular boundaries, as if the metal has been plucked apart.

A more likely explanation for the spongy zone is that it represents a region of compromised metal that has fractured through void coalescence when the existing stress-crack was widened by the blow of the hammer. Exactly how we might end up with a region of compromised cohesion is something I will consider in a later post.
I was curious to understand how much of what can be observed at the fracture surface is related to corrosion processes occurring below the surface of the plane of the fracture. To this end I ground the fracture surface back by approximately one millimetre, polished it and then subjected it to a brief etch in H20/HCl/HNO3, 10/10/1. The result can be seen in the photo below.

Not only did this process reveal axially-oriented stress-cracks underlying the original crack surface, but also, and far more intriguingly, it revealed a field of what appear to be true anodic dissolution tunnels. This feature would appear to be something quite different to that observed in the spongy fracture zone of this bolt.
The red arrow in the figure above marks the direction of the progression from stress-crack to brittle fracture that was observed in the millimetre or so of metal that overlay this newly-prepared surface. The four photos below show what happens as we progress from the part of the bolt underlying the stress-crack surface proper, to that underlying the brittle fracture surface.




We’ll talk about those tunnels in a minute.
Firstly, let’s be perfectly clear what we are looking at here. This is not a fracture surface as such, but a flat surface that I ground and polished a millimetre or so below the plane of the original fracture. It should be flat, and if it is otherwise, then, that is the action of the etchant.
There is a tendency to forget that when etchants and colorants are applied, what you see is the result of a chemistry experiment, and that care is needed to map the picture back to the real world.
This etchant preferentially attacks the inter-granular boundaries of austenitic stainless steels and thus brings the metal grains into physical relief. You can see this very mild etching action in the last photo of the sequence. It is in marked contrast to the first of the sequence where substantial amounts of material have been removed without selectivity for the inter-granular boundary. This fact alone tells us that the metal under the stress-crack fracture zone differs in some way to that under the brittle fracture.
There are two features that are abundant in the metal under the stress-crack fracture zone, that are very much less well represented beneath the brittle fracture zone. The first is the occurrence of greigite. It is clear greigite is resistant to the etchant, and it is increasingly revealed as the base metal is etched away. The second is the abundance of tunnel structures. Regardless of the role the etchant may play in revealing these structures, the fact remains that there are many more of them to be found under the stress-crack fracture zone.
A further significant observation is the correlation between tunnel diameter, greigite occurrence and the susceptibility of the metal to the etchant. This finding makes it looks like we are seeing progressive stages in the degradation of the chemical/mechanical properties of the metal.
Thus we might postulate that the brittle fractures we observe are little more than indicators of the early stages of a process that ultimately leads to stress-cracking and total loss of integrity.
So are SRB responsible for ‘eating’ those tunnels? Right now, I’d reckon that this is the most likely explanation for what we are seeing. Confronted with a lack of a suitable organic substrate, the bacteria switch their metabolism over to one of direct electrical connection to the metal surface, thereby releasing metal cations, i.e. dissolving the metal surface, as they pass the extracted electrons down their metabolic chain to the ultimate receptor which is sulphate .
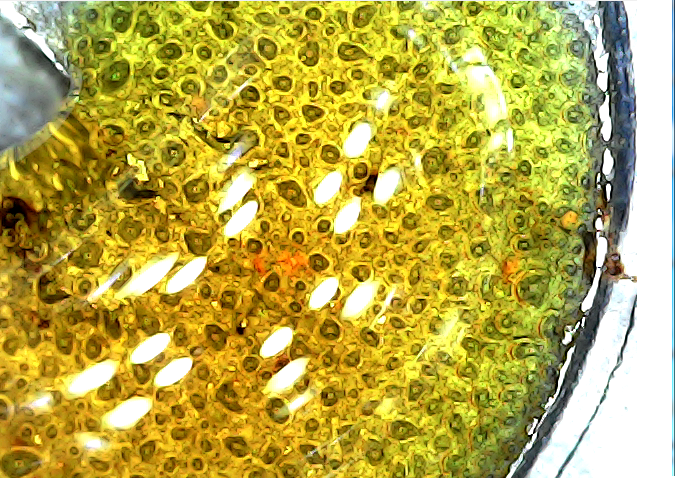
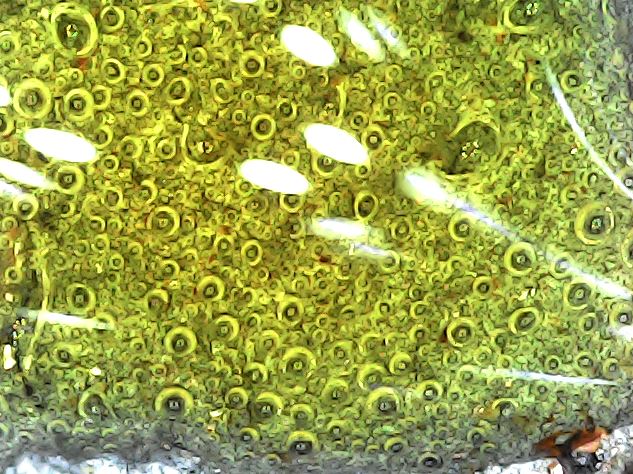
If so, we should see the bacteria if we look inside those tunnels – right? Well yes, but that is surely easier said than done. However, being ever the optimist, I decided to take a look with my metallurgical microscope – it has the advantage of a confocal illumination system that shines a light wherever it looks, so as to speak. To my great surprise it didn’t take me long to come up with the images shown below.



Let’s start with some reality checks. A typical SRB like Desulfovibrio vulgaris is some 2um to 3um in length. The metal grain size of the specimen is pretty variable, but 10um to 30um would be representative. The orange and yellow objects that can be seen adhering to the surface are 10um or so in size, and are clearly clusters of smaller objects. The silver-grey surface is easily interpreted as the deeply-etched metal, but its finely spongiform surface, one that exhibits some sort of granularity of the order of 1um, is a matter for conjecture.
We can be reasonably confident that we are looking at SRB in situ in these images, but further verification would be nice. To this end I took a longitudinal section, the plane of which cuts through the transverse section we have just been discussing. It roughly follows the path of the red line in the figure below.




Once again it is important to stop, and clarify what we are looking at here. The surface showing in the figure above is not a fracture surface, but a cut I made to reveal structures hidden below the original fracture. It is a perfectly-flat, ground-surface that is intersecting corrosion features within the solid metal. I have then taken a further section vertically into this surface to give a new view that intersects along the red line.
In the figure above I show images representative of three zones along the the intersection of the two planes labelled A, B and C. The straight edge of the intersection can be seen on the right of the sub-images. For Zone A, the line of intersection is substantially coherent given the lack of corrosion tunnels in the metal below the brittle fracture surface. However, moving to Zone C, which lies below the spongy fracture surface, the straight edge is less obvious due to the deeply-tunnelled and fractured nature of the metal at this point.
I’ll note in passing that, although the section at Zone A is largely free of intersections with tunnel features, changes to the material of the \delta-ferrite stringers do seem to be occurring. Further to this I’ll note that in the transitional zone B, there can be seen short, straight tunnels that seem to have initiated at locations of \delta-ferrite. We will come back to consider this when we start to look at the possible mechanisms in play. Parking for later discussion some of the interesting observations that could be made, I want to come back to the current focus of this post, which is the penetration of solid metal by SRB themselves.
Taking a closer look at Zone C provides some intriguing observations. Here we see strong evidence for bacterial colonisation of the interior of the bolt.
Note: In the images that follows we are dealing with a polished longitudinal section devoid of any etchant or colorant. Any feature that stands out in contrast is either a physical crack, or a chemical alteration caused by the SRB environment.
Within Zone C, the metal matrix is extensively fractured. In the image below, we see cracks at all stages of formation, from incipient lines within relatively solid blocks of metal, to cracks some microns wide. The pattern of the incipient cracks is something that will come up for discussion in later posts. The sides of the cracks are relatively smooth, or they are until the SRB get to work on the metal exposed by the cracking process.

And, it doesn’t take much searching to find bacterial cells associated with cracks exhibiting heavily sculptured sides. In the image below we see a large clump of bacteria tightly packed into a crack. Given they are stacked partially end-on, their apparent size is somewhat reduced, and, after making allowance for that fact, I’d judge them to be about 1um in length.

This dimension is at the lower end of the size range recorded for a common SRB such as Desulfovibrio vulgaris, but is by no means unusual, as the image below taken from confirms.

Although it is reassuring to make this sort of morphological comparison, in truth, without genetic sequencing, any guess we might make is likely wrong.
The sculpturing that occurs as the SRB widen the crack by dissolution of metal is astonishing in its fine detail. The image below is typical, with bacterial cells (small dots) lining the finely-structured incursions into the metal matrix.

A somewhat closer view reveals the intricacy of the dissolution pattern.

The very fact there is a pattern of metal removal is intriguing, and surely points to underlying structural cause.
The image below reveals a marked cross-hatch pattern.

It seems likely that this pattern reflects underlying slip bands within the crystal lattice. I will elaborate on the significance of this finding with respect to the localisation of strain-induced ∝´-martensite, and the conduction of atomic hydrogen through the metal matrix when we consider the atomic aspects of hydrogen embrittlement in a later post.
5. The Mechanism of SRB Attack:
In the past few years significant advances have been made in the understanding of the behaviour of SRB at the molecular level. This is a big subject, and one that could easily occupy multiple posts. For now, I will do little more than attempt an overview of the basic principles, and see how they align, or not, with our observations.
I will elaborate on just two of the many possible redox reactions that I think are likely to be critical to the SRB attack we are seeing at Cabo da Roca.
The first of these is the anodic process whereby we see the removal of metallic iron, and, as such, it represents a mechanism by which the bacteria could gain entry into the interior of the bolt.
The second is the cathodic process by which hydrogen ions are reduced to atomic hydrogen on the surface of the metal. Although not corrosive as such, it is important because of the potential to sponsor hydrogen embrittlement.
The two process are illustrated below. Note that in the case of the anodic process, the bacteria are considered to be playing an active role in removing metal from the surface of the bolt, whilst the cathodic process is merely a consequence of the environment. However, the above distinction is perhaps moot because, in the final analysis, all anodic metal loss is driven by the extraction of electrons from an electrically connected surface exposed elsewhere in the aqueous environment. The end result is the same regardless of whether it is bacteria extracting the electrons, or the reduction of hydrogen ions at a locally cathodic surface,

a) Anodic Process: Bacterial cells electrically-connect their metabolic, electron-transport chain to the metal of the bolt, thereby utilising the redox potential of the Fe0 / Fe++ couple to reduce sulphate to sulphide, and, in the process, derive the metabolic energy necessary for subsequent cell division and colony growth. Metal is dissolved.
b) Cathodic Process: Hydrogen ions accept electrons from the metal surface to leave atomic hydrogen absorbed to that surface. Some hydrogen diffuses into the metal, whilst a limited amount recombines at catalytic sites on the metal surface to release hydrogen gas. Diffusion of atomic hydrogen into the metal gives rise to embrittlement.
The Anodic Process:
If our corrosion story hinges on something rather special and unusual, then this is it. As I wrote in Part 1 of this series –
SRB are widely-distributed organisms in swampy, low-oxygen environments where there is an abundance of organic matter to provide the reduction potential that fuels their metabolism. In analogous terms to a fire, the organic matter is the wood, and sulphate is the oxygen. It is therefore remarkable to find such an organism occupying a niche on a barren cliff that is free of organic matter. It seems that there is a very special adaption that lets this organism swap over to direct, electron-transport reduction when starved of organic substrate. Even given the abundant sulphate, and the availability of anoxic crevices in the rock, survival would be a struggle until such time as a climber comes along and places a bolt. The bacterium literally plugs into the bolt and thrives as it “burns” metallic iron in place of organic matter. And as SRB thrive, they create a sulphide rich chemical environment that radically alters the typical corrosion processes at play in the marine cliff environment.
This phenomenon of “plugging-in” to a metal substrate is known as EET (extra-cellular electron transport). As a mechanism, it goes a long way in explaining why an SRB environment can often be benign, or even protective, from a corrosion standpoint, yet under certain circumstances may become very aggressive, with an attack rate of many millimetres of metal per annum. Satoshi Wakai, provides a nice historical overview of the research area that has become known as electro-microbiology.
For readers with a bit of biochemistry knowledge, the figure below from Deng and Okamoto,, should help explain this very cool phenomenon.

Provided sufficient sulphate is present in the environment, the thermodynamics are such that metal ions will move into solution as electrons are withdrawn through the electrically conductive connection of the bacterium. In turn, the flow of electrons though the bacterium supplies it with the chemical energy necessary for cell division, and, as new bacterial cells are formed, they attach to the newly exposed metal surfaces. Thus, it is not hard to see how these bacteria can “drill” their way into solid stainless steel. The fact that individual cells are physically attaching to the metal surface before corrosion can be initiated, goes some way to explaining the finely sculptured nature of the corrosion surface we see under the microscope. The fine relief of the surface matches the observed size of the bacterial cells.
What are the limitations to the anodic process?
As we have just noted, the energetics are totally dependent upon there being sufficient sulphate ions in the liquid bathing the bacterial cells. Clearly, there is no shortage of metallic iron and, given there is no sulphate available within the rock itself, it is obvious that all sulphate has to come from the external environment. Surely the torturous pathway from the rock face to the bolt surface beneath the fixed hanger, and then through micron sized corrosion tunnels to the bacteria themselves, is diffusion limiting?
Yeah, nah, maybe, maybe not. On one hand we can point to the long time scales over which the process is unfolding, along with the fact that the mass transport requirements are not so large given the stoichiometry of four ferrous ions released for every sulphate ion consumed, all of which suggest that sulphate supply need not be diffusion limited. On the other hand, why is it that we seem to need very high sulphate levels at a sea cliff before we observe SRB corrosive attack on the stainless steel infrastructure? It really does seem that exceptional sulphate levels are required to drive exceptional corrosion by SRB.
The fact that we don’t see SRB attack when external sources of sulphur are absent, cautions against a simple diffusion constraint hypothesis. After all, the level of sulphate in sea water is not insignificant, and, it is well documented to be a driver of SRB corrosion in environments such as the mud of the seafloor . Yet, worldwide wall-wash sampling indicates that, for a sea cliff to be corrosive, sulphate levels need to match that of chloride, as illustrated by the graphs below.

It almost seems as if there is a threshold sulphate level that must be crossed if SRB attack is to be observed. As striking as this observation is, I’d hesitate to make such a subjective assessment given how little we know. As I will detail below, there are some serious contradictory aspects to the evidence on hand.
The anodic process presents a stoichiometric enigma:
We have a problem in that the anodic process should lead to an ever-increasing alkaline environment given the stoichiometry of eight hydrogen ions consumed for every sulphate ion that is reduced.
{\scriptsize4Fe^0 + SO_4^{2-} + 8H^+ \longrightarrow FeS + 3Fe^{2+} + 4H_2O~~~~~Eqn.~1}Not only that, but it should be noted that just one third of the iron consumed ends up being associated with the mono-sulphide. And, by analogy, even less would be associated with greigite and pyrite. What happens to the balance? Surely, an inescapable consequence of such stoichiometry would be that the dominant corrosion product cannot be iron sulphide, whatever form it might take?
If we turn aside briefly to look at what is known about other SRB environments, then provide solid evidence for an alkalising process coupled with the uptake of environmental bicarbonate to sequester the iron not coupled to sulphide as the iron carbonate, i.e. siderite.
{\scriptsize 4Fe^0 + SO_4^{2-} + 3HCO_3^- + 5H^+ \longrightarrow FeS + 3FeCO_3 + 4H_2O~~~~~Eqn.~2}This equation implies the SRB metabolic processes are running within a buffered carbonate/bicarbonate system, and thus we can anticipate neutral to mildly alkaline conditions to prevail.
From the same paper, the figure below illustrates the thick encrustation of corrosion products, and the mode of corrosion typical of this type of SRB attack.

The authors postulate a mechanism whereby, under the neutral to basic conditions of the prevailing HCO3– /CO32- buffer system, mackinawite/siderite deposits will coat the metal surface, and thus block bacterial access. Instead of connecting electrically to the metal surface, the bacteria connect via the semi-conducting sulphide coating. The authors postulate that the ferrous ions released by the anodic process reach the exterior surface by a combination of diffusion and direct hydraulic connection.

This situation is quite different to that we are observing with the bolted infrastructure of Cabo da Roca. If we consider just the corrosive, anoxic, inner domain of the anchor we can make the following comparison.
| Feature | This study | Enning et al |
|---|---|---|
| pH of the environment | 2.0 – 3.5 | 7.0 – 8.5 |
| solid corrosion products present | Fe3S4 only | 1FeS +3FeCO3 |
| degree of coating of the metal surface | none | extensive, thick |
| direct bacterial attachment to metal | yes | no |
| nature of metal removal | penetrative | superficial |
It is clear that the potential of bacteria to “drill” narrow tunnels deep into the metal can be realised only under conditions sufficiently acid to ensure that sulphide deposits do not occlude the transport of sulphate into, and ferrous ions out of such narrow spaces. Furthermore, the micron-scale surface sculpturing of the corrosion surface would be possible only if the bacteria could directly connect to bare metal, rather than through millimetre-scale sulphide deposits.
Aside from the stoichiometric difficulties with Eqn. 1, all evidence points to the existence of a unique, acidic SRB environment which is the enabler of the aggressive, penetrative corrosion we see at Cabo da Roca.
However …
A unique acidophilic SRB it might be, but the thermodynamics still need to make sense:
There are distinct, thermodynamic problems in postulating a low pH environment that is also strongly reducing. It is unstable in the same sense that placing an iron nail in dilute acid is unstable. The iron, a strong reducer, will dissolve, and the acid will be neutralised with the release of hydrogen gas. Let’s explore this idea a bit.
The natural environment is subject to the laws of thermodynamics, and thus, it is not surprising to find that systems which might range from natural waters and soils, to the micro-organisms inhabiting such, all shun reducing environments of a low pH. have collated some 6,200 data points from the published literature to demonstrate the limits of natural systems within the redox-potential / pH space. The diagram below illustrates the situation they found for SRB. It can read in an analogous way to the Pourbaix diagram used in corrosion studies.

The authors use physico-chemical reasoning to place bounds within the redox-potential/pH space for the operation of natural processes. These bounds are represented by the solid black line. It is not that processes can’t exist outside of these bounds, but rather, from the perspective of the authors’ field which is concerned with geological times scales, such processes are unlikely to have sufficient stability to impact the subsequent geology.
The authors map into this space all the examples of SRB isolates they could locate in the literature, and yes indeed, they are contained by the proposed bounds for the redox-potential/pH space, and yes, as would be anticipated for the dissimilatory reaction of Eqn. 1, they are clustered at the reducing, high-pH corner of the space.
I’ve added to the diagram a blue line that represents the equilibrium boundary for the reduction of H+ to H2, as per Eqn. 3 below.
{\scriptsize 2H^+ + 2e^- \longrightarrow H_2~~~~~Eqn.~3}It can be seen that the bounds laid out by the authors are situated above this line, and thus the natural environment rarely encompasses conditions of sufficient reduction potential for hydrogen gas to be evolved. This proposition makes sense in terms of stability on geological time scales. Of course, Man can, and does, violate these bounds, through activities such as burying an iron pipe in marshy ground. Under such circumstances we might well see hydrogen being evolved, but the event would be of such trivial magnitude and duration as to be insignificant in the face of long-term geochemical processes.
In the process of assembling this admirable overview of natural processes, the authors’ take a swipe at fellow researchers within their field that ignore physical chemistry in pursuit of a good narrative. As a grumpy old dude with an overdeveloped sense of respect for classic science, I’m 100% onboard with that sentiment, so, what am I to do about the fact that the current study at Cabo da Roca seems to be positioning us at the unlikely location I’ve circled in red, in the redox-potential/pH diagram above? I’m happy enough with the idea that we have bumbled across something unique, but stop well short of attributing it magical properties. We need to keep digging. Thus far what we have isn’t making a lot of sense.
How certain are we that the environment is acidic?
There is no doubt that very acidic conditions exist within inner regions of the bolt structure. The evidence from simple pH paper tests is overwhelming, and whilst I want to stay closely focused on Cabo da Roca (we don’t need to go looking for extra complexity!) I will note that we have observed the same phenomenon at the corrosive sulphate cliffs of Railay in Thailand.

Furthermore, we have a pretty good handle on the major electrolytes of the system because we have measured them. On the basis of these measurements we need to factor in sodium, calcium, chloride and sulphate. Thus at low pH we are dealing with a mix of hydrochloric, and sulphuric acids with little in the way of buffering. The fact that the acid substances on the surface of the anchor components are readily rinsed-off supports the above assertion.
However, what is meant by “inner” needs to examined. By laying moist pH paper across sections of a disassembled anchor, strikingly sharp transitions from low to high pH are observed at points such as where a washer was covering a fixed hanger, or a nut was covering a washer and so forth. The low pH is always on what was the “inner” surface of the intact assembly.
Given that those “inside” regions where we measure strong acidity are very much toward the “outside” because there are distinct limits to what can be done with a piece of pH paper, just how safe is it to assume that acidic conditions persist right into the innermost regions colonised by the bacteria? After all, we have every reason to believe the bacteria will be alkalising their environment as they multiply.
Fortunately, we have another line of reasoning we can bring to bear on the problem. We know from microscopy that all evidence points to the metal surfaces being free of sulphide with the exception of pockets of greigite crystals. It is only at the points of transition to “outside” do we observe metal surfaces coated with mackinawite. Proceeding even further to the “outside”, and onto the external rock surface, it is not uncommon to find thin deposits of rust. Although it is a hallmark of the brittle fracture phenomenon that external signs of corrosion are minimal, or indeed absent, nevertheless, all bolts if left long enough begin to show signs of rust seeping out from internal spaces.
I have annotated the Fe/H2O/H2S Pourbaix diagram of to illustrate the fact that, given the evidence for sulphide formation, we can draw an upper limit on the pH of the environment within the SRB corrosion tunnels.

We can see that for mackinawite to be excluded, the pH has to be less than 4. Notice also, that at this pH, there is the possibility of greigite formation in the absence of mackinawite as the environment becomes less anoxic.
Taken together, the pH paper evidence plus that from observing the distribution (or not) of sulphides, lends considerable weight to the idea that the SRB environment is indeed acidic.
However, the sceptic in me cautions that inferences drawn from Pourbaix diagrams are only as good as the input data. For starters, we need to recognize that the concentrations of Fe++ and H2S within our system are unknown, and, to all practical purposes, unknowable with any exactness. The diagram above shows that the effect of increasing Fe++ is to shift the mackinawite transition to lower and lower values of pH. On this basis a lower limit of pH 4.0 seems conservative given I would expect ferrous ion concentrations to be in excess of 100ppm.
If we are underestimating the ferrous ion concentration by maybe an order of magnitude, the opposite is likely to be true for H2S. The authors constructed the diagram for a partial pressure of 0.24 bar. Applying the Henry’s Law constant for H2S, we find this partial pressure is equivalent to 47mM which is is something like an order of magnitude higher than that which would normally be considered inhibitory for SRB metabolism . Maybe we do have a unique SRB in terms of its resistance to H2S toxicity, but I’m inclined to go with Occam’s Razor and assume that the real world value of H2S partial pressure is an order of magnitude lower than that used as the basis of the Pourbaix diagram in question. Correcting for Fe++ concentration would shift the pH transition point down to say pH 3.5, whilst dropping the H2S partial pressure would shift it up to say pH 4.5. On balance then, assuming a pH <4.0 seems the safest estimate we can make based on the observed absence of mackinawite deposits.
But, there is more to stainless steel than just iron:
Up to this point in the discussion, we have restricted our analysis to the equilibrium conditions governing the iron sulphides. However, austenitic stainless steels, such as 304 or 316, contain approx. 18% chromium, and thus the equilibrium behaviour of chromium in the sulphide system ought not be overlooked.
It turns out that chromium sulphides are not stable under any of the conditions we are likely to encounter. Furthermore, because of the prevailing anoxic conditions, formation of a passive chromic oxide film cannot occur, and an equilibrium between the chromous ion and chromium metal will persist across a wide range of neutral and acidic conditions. This is illustrated in the Pourbaix diagram below, taken from .

So it looks like chromium dissolution will occur under the pH regime we are proposing without issues of precipitation of insoluble corrosion products. Can the same be said about the 8% to 10% nickel content?
The Pourbaix diagram below, taken from the same paper, indicates that the pH conditions for mono-sulphide are broadly similar to that for iron, and thus we can take the absence of sulphide coating on the bare metal as an indication that the pH of the environment must be less than 4.0. The diagram was constructed for a H2S concentration of 15uM and a Ni++ concentration of 1ppm, which as per preceding comments on the iron sulphide system supports the idea of an upper pH limit of 4.0

Thus, based on the known behaviour of both the nickel and the chromium sulphide systems we have no cause to modify the pH estimate we argued is likely the case for iron.
Are acidophilic SRB a known thing?:
There are good theoretical reasons to believe that most SRB will favour neutral to alkaline environments, and earlier, I cited several studies that support this idea. Thus, if the environment within the Cabo da Roca bolts is indeed acidic, how likely is it that we have stumbled across an acidophilic SRB?
A line of reasoning that bears upon the question of uniqueness is that, as far as we know, all bacteria with SRB capability attain that characteristic by virtue of the same archaic DNA sequences. That is, all known SRB employ the same dissimilatory sulphur metabolism described by Eqn 1, and thus are likely to favour the neutral to alkaline pH range.
Against this contention I’d raise the old truism, “absence of evidence is not evidence of absence”. It may be that the preponderance of alkaline SRB systems reported in the literature is little more than a bias introduced by the commercial importance of “buried pipe” corrosion to the oil and gas industry. These are organic matter rich environments and distinctly different to the one we are studying.
Indeed, it looks like the growing importance of bio-remediation of acid mine drainage might shed a new light on the matter. For example, report SRB isolates from mine drainage capable of functioning at a pH of less than 2. provide an up-to-date review of this subject, and the possible mechanisms by which SRB adapt to extremes of pH.
Thus it seems that there is no sound basis on which to reject the possibility that we are dealing with an acidophilic SRB. Such organisms evidently do exist.
How toxic are Cr and Ni to SRB?
Continuing down our path of obdurate scepticism, the question arises as to how well a sulphate-reducer is going to tolerate the chromium and nickel levels that must be released by the anodic process. Recall that of the metal ions released, we can anticipate some 18% will be chromium – perhaps a mix of Cr2+ and Cr3+ but not Cr6+ – while 8% to 10% will be Ni2+. Recall also that we have just argued that none of these ions are going to drop out of solution because of the acid conditions, and thus, if they are toxic, will be fully available to the bacteria.
So how toxic might these elements be? Well, for starters, we don’t know their concentrations. For the purposes of the earlier equilibrium analysis via Pourbaix diagrams I chose 100ppm for iron and proportionately less for chromium and nickel. If, for the sake of the argument, we stay with these concentrations then we would be looking at 20ppm for chromium and 10ppm for nickel.
A study by provides the following inhibiting concentrations, Cr(III) 60ppm and Ni(II) 20ppm. Those figures are very much in our ballpark, so what are we to make of this? Could any ordinary SRB attack stainless steel before self-inhibiting?
A closer look at the paper reveals substantial system complexity behind these bald figures. There are many factors at play, an awareness of which should caution against crude extrapolation, yet, other studies seem to end broadly at the same point, with nickel inhibiting in low ppm range and chromium in the upper range.
Direct comparison is fraught due to differences in pH and redox potential between these experimental systems, not to mention likely differences in the organisms themselves. If anything, it looks like the concentration of iron in solution has to be much less, perhaps an order of magnitude less, than the 100ppm we proposed earlier, if nickel levels are not to be totally inhibitory. Either that or we are dealing with a SRB that is not only unusual as an acidophile, but also is unusually nickel tolerant.
The cathodic process:
Given that we are certain acidic conditions exist, and given that the metal surface is known to be free of sulphide deposits, the inescapable conclusion is that we must have metallic iron in contact with a solution of sufficient hydrogen ion concentration to drive the cathodic production of hydrogen gas at its surface.
In the figure below, I have annotated the Fe/H2O/H2S Pourbaix diagram we were considering earlier to show the equilibrium boundary for the reduction of H+ to H2 in red. It is clear that the reduction potential exists to drive cathode production of H2

The above consideration begs a question I have alluded to previously, and that is, why don’t we see the aggressive dissolution of metal accompanied by the production of hydrogen gas? I believe the answer is that the reaction is inhibited for the same reason that a small chip of sodium sulphide will completely suppress the formation of hydrogen bubbles at the surface of an iron nail immersed in dilute acid. The suppression takes place with no visible formation of a sulphide coating. Let’s take a look at what might be happening.
I have already introduced the basic cathodic reaction back up the page, but to avoid scrolling, I will write it again –
{\scriptsize 2H^+ + 2e^- \longrightarrow H_2~~~~~Eqn.~3}This classic reaction is understood to be made up of two distinct processes. The first is the reduction of hydrogen ions at the metal surface to produce atomic hydrogen absorbed at that surface, while the second involves surface diffusion of the atomic hydrogen so formed to catalytic sites where recombination to molecular hydrogen occurs.
It is the second process that is rate limiting and subject to poisoning by hydrogen sulphide. The figure below is taken from . These authors make the interesting observation that substances that poison industrial, catalytic, hydrogenation processes are the same ones that poison electrolytic, hydrogen recombination.

The authors argue that the effect of poisoning of the catalyst is to reduce both kr and kd proportionately such that the recombination/dissociation equilibrium is unchanged. This proposal is in keeping with the notion that there are only so many catalytic sites distributed across the surface, and the blocking of a number of these with a poison such as sulphur will increase the mean distance atomic hydrogen must migrate to achieve recombination. If the density of catalytic sites falls low enough, the recombination/dissociation reaction will all but cease.
Suppression of the recombination reaction ultimately suppresses the reduction of hydrogen ions back upstream, and beyond that, any coupled anodic process that might dissolve metallic iron is likewise suppressed. Thus given sufficient dissolved hydrogen sulphide, we can expect hydrogen evolution and metal dissolution to cease, but, and this is a critical point, we are still dealing with a system in equilibrium and thus would anticipate that the surface would become saturated with respect to atomic hydrogen. Under these circumstances we can anticipate substantial rates of diffusion of hydrogen into the metal itself.
It can be seen that an important consequence of the acidic, reducing conditions in the presence of hydrogen sulphide will be to sponsor high surface concentrations of atomic hydrogen, and thus set the scene for significant hydrogen migration into the metal, and its subsequent embrittlement.
In summary, it is clear that we are dealing with an acidophilic SRB that, by virtue of the acidic environment, and the EET mechanism, is able to tunnel deep into, and subsequently colonise, the metal of the bolt. There are no substantive objections to this explanation that can be raised on grounds of electrochemistry or microbiology.
However, we are yet to explain how such an acid environment is maintained in the face of the strongly alkalising, dissimilatory reduction of sulphate to sulphide.
6. One Question Remains Unanswered:
Remember Eqn.1 which we discussed back up-page? This is the equation for the dissimilatory reduction of sulphate to sulphide using metallic iron as the electron source.
{\scriptsize4Fe^0 + SO_4^{2-} + 8H^+ \longrightarrow FeS + 3Fe^{2+} + 4H_2O~~~~~Eqn.~1}This equation tells us that, for every atom of iron dissolved, we dispose of eight hydrogen ions and thus, inevitably, must increase the pH of the environment. Yet, as we have just discussed, the pH has to be kept below 4.0 if the bacteria are to maintain their unique penetrative mode of attack.
Until we can rationalise this enigma, we really won’t have the full story.
One important clue as to what might be happening is the observation that, just as “inside” surfaces can be very acidic, so it is that outside surfaces can be very alkaline. An example of this situation is illustrated below. In this particular case we see zones of approximately pH 1 to 2 in very close proximity to zones of pH 9 to 10. If we look at the surfaces in question, it is clear we are dealing with the transition from the oxygenated outer surface to inner anoxic regions created by the contact between the washer and fixed hanger. Notice also the somewhat thicker layer of corrosion products toward the inner region.

Apart from telling us that we have very different chemical environments in close proximity, comparing the visual appearance of the surface with the pattern of pH differentiation doesn’t really add much. However, judging by the location of the corrosion products, it does seem likely that this example would have exhibited the so-called “black ring of death” along the line of the exposed contact point between washer and fixed hanger.
The term “black ring of death” was coined by John Byrnes some twenty years ago to describe what he saw as a warning sign of internal weakness among bolts at Cayman Brac. The phenomenon has since been recognized at other ‘sulphur’ crags throughout the world. The picture below provides a nice example.

It should be pointed out that the development of dark corrosion products need not be as well developed as in this example from Kalymnos. In cases of what I assume to be earlier stages of development, the presence of corrosion products often are barely discernible, yet the bolt can be dangerously embrittled.
Furthermore, the pH differentiation and deposition of corrosion products is sometimes confined to the space between the fixed hanger and the rock, rather than between washer and fixed hanger. The bolt pictured below is from Ibiza, and serves as a good example of such development.

The lower surface of fixed hangers can exhibit very alkaline conditions as the examples pictured below illustrate.

Is the “black ring of death” a barrier biofilm?
Based on the evidence before us, we know one thing for sure. A physical barrier of no more than 1mm thickness is partitioning two very different environments – an inner, anoxic, low pH region and an outer, oxygenated, high pH region.
It is unlikely that such a partitioning is thermodynamically stable, and therefore it is reasonable to assume that what we are observing is being maintained by a metabolic process. By such a reckoning, our physical barrier needs to be a living biofilm. I have illustrated this hypothesis below.

So far so good, except that having previously argued that the paucity of complex carbon substrates is a unique feature of the rock anchor environment, we need to be looking for a chemoautotrophic process as our energy source.
One obvious possibility is the oxidation of Fe2+ to Fe3+ as oxygen becomes increasingly available across the biofilm barrier. IOB (iron oxidising bacteria) are ubiquitous organisms that occupy such niches in the environment where ferrous ions are flowing from anoxic to oxygenated spaces. Surprisingly, given that their discovery dates back into the 19th century, very little is known about these organisms because they are difficult to establish in laboratory culture. See for a recent review. They catalyse the typically slow oxidation in air of the ferrous to the the ferric state as per Eqn. 4, extracting metabolic energy from the process as they do so.
{\scriptsize 4Fe^{2+} + O_2 + 10H_2O \longrightarrow 4Fe(OH)_3 + 8H^+~~~~~Eqn.~4}They also fix carbon from carbon dioxide to sustain cellular growth, and, significantly from the point of view of biofilm formation, exude a large part of the acquired carbon an exopolysaccharide.
Although we are well and truly venturing into the realms of speculation here, it is worth noting that IOB working in consort with the SRB would solve the stoichiometric enigma presented by SRB as the sole agent of corrosion.
It provides a sink for the excesses in both in Fe2+ and OH– we previously commented upon. I have illustrated the idea below.
It is relatively easy to locate talk within the published literature of biofilms allowing different species to act in consort, and in doing so to exhibit unique characteristics such as enhanced corrosion rates. However, moving from such hand-waving to rigorous elucidation of form and function seems to be problematical. To my mind, laboratory experiments are poor replicas of the natural environment and leave much to guesswork.
Nevertheless, the idea that we might be looking at SRB working in consort with IOB is intriguing, but, I’d hasten to add that without positive identification of IOB within the “black ring of death”, and without definitive chemical analysis, it should be regarded as highly speculative.

7. After all that, what can we conclude?
Well, after such a time-consuming deep dive, what do we have to show for the effort? I believe the following conclusions to be solid.
- The observed brittle-fracture phenomenon results from colonisation of the solid metal of the bolt by a novel, acidophilic SRB.
- The form of the attack is novel in that it involves penetrative corrosion under conditions sufficiently acidic to suppress sulphide deposition at the active surface. This differs from the better known circum-neutral process of superficial corrosion beneath a thick deposit of mixed sulphide and carbonate.
- The form of attack sponsors high levels of atomic hydrogen at the surface of corrosion tunnels within the solid metal. Subsequent hydrogen embrittlement is almost certain.
- The stoichiometry is incomplete without the agency of coupled oxidative process operating at the interface with the outside air. It is suggested the commonly observed “black ring of death” is a biofilm providing such an agency. This hypothesis is as yet unproven.
Coming Next:
In the next part of this series I will turn my attention back to the brittle fracture phenomenon proper, and will discuss how what we find there aligns with current understanding of hydrogen embrittlement.
Discover more from Crag Chemistry
Subscribe to get the latest posts sent to your email.
One reply on “Corrosion at Cabo da Roca – 4”
With regard to #17 Dennis Enning (2012) – there is a new paper authored by him and Julia Garriefs in 2014.
Link: https://journals.asm.org/doi/full/10.1128/aem.02848-13
Interesting read…